close friends and relatives know what to do if this occurs. It is also advisable to carry or wear something stating that you are being treated with insulin.
If you lose consciousness due to hypoglycaemia your relatives or close friends may have been taught to give you a subcutaneous or intramuscular injection of glucagon. You should always take sugar or glucose on recovery. If you do not respond to glucagon or it is not available, your relatives and close friends should know to call a doctor immediately so that intravenous glucose can be given.
Hyperglycaemia (high blood sugar)
This results from taking too little insulin in relation to your food intake or exercise. If your blood sugar is high you are likely to be very thirsty and will need to pass urine frequently. Prolonged high blood sugar may eventually lead to coma. If your blood sugar level rises to high levels check your urine for ketones. If ketones are present contact your doctor immediately.
Side effects
Side effects to insulin are rare. The most common is itching or redness at the site of injection. If you experience any skin reaction at the injection side, tell your doctor as soon as possible.
When insulin treatment is first started you may get swollen feet and blurred vision in the first week or so. These problems usually disappear with continued treatment; tell your doctor if they persist.
Changing to human insulin
You should only change to human insulin on your doctor’s advice.
Transfer from porcine to human insulin of the same type, does not normally require a change in dosage.
Transfer from bovine or mixed bovine/porcine insulins to human insulin may require a dosage adjustment; follow your doctors advice on this.
A few patients have reported that after being transferred to human insulin, the early warning symptoms for hypoglycaemia were less pronounced than they were with animal source insulins.
***Storage of disposable pen preparations
As with all insulin preparations, store your full packs of disposable pens, and pens not in use, in a refrigerator at 2° to 8°C, but do not allow the insulin to freeze.
Pens which are in use must not be kept in a refrigerator. Pens in use or carried as a spare can be kept at normal surrounding temperature (e.g. in the pocket or handbag) for up to one month, but should not be exposed to excessive heat or sunlight.
What's in your medicine
Human Mixtard 30 Pen contains 3 ml of a suspension of human insulin (pyr) in a strength of 100 iu/ml. The suspension appears white and cloudy when shaken. Human Mixtard 30 Pen also contains glycerol, sodium phosphate, protamine sulphate as retarding agent and m-cresol and phenol as added preservative.
Important information for users of Human Mixtard 30 Pen
The product was formerly called PenMix 30/70.
Do not use any of the products of Human Insulatard or Human Mixtard 30/70 with dark blue or dark pink packaging respectively. This is older stock which contains Human insulin (emp); these products were discontinued in April 1995.
If you used Human Insulatard, or Human Mixtard 30/70 prior to April 1995 who should follow your doctor’s advice with regard to dosage change on transfer to Human Mixtard 30 Pen. Monitor your blood glucose more carefully for some time after transfer; diabetic care may need to be increased during this period. If you notice changes in your usual range of blood glucose requirements during this time you should discuss this with your diabetes specialist.
Instructions for use
Please read these instructions carefully before using your disposable Pen.
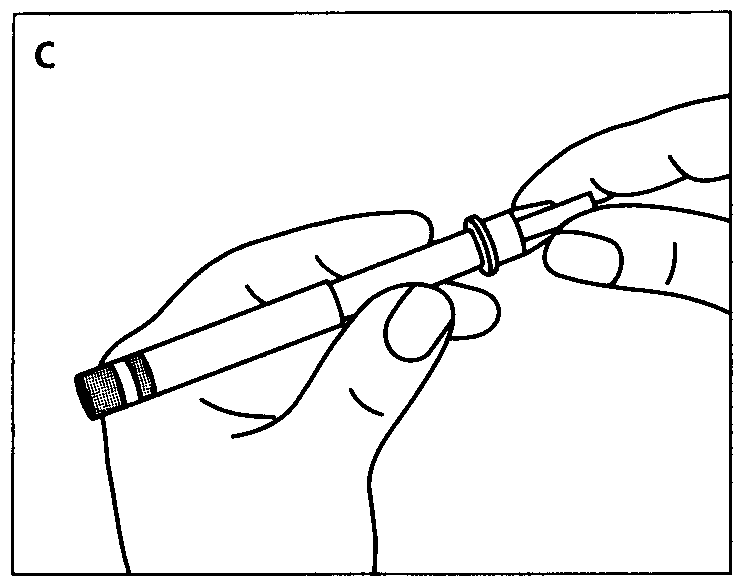
Human Mixtard 30 Pen must only be used with NovoFine® needles.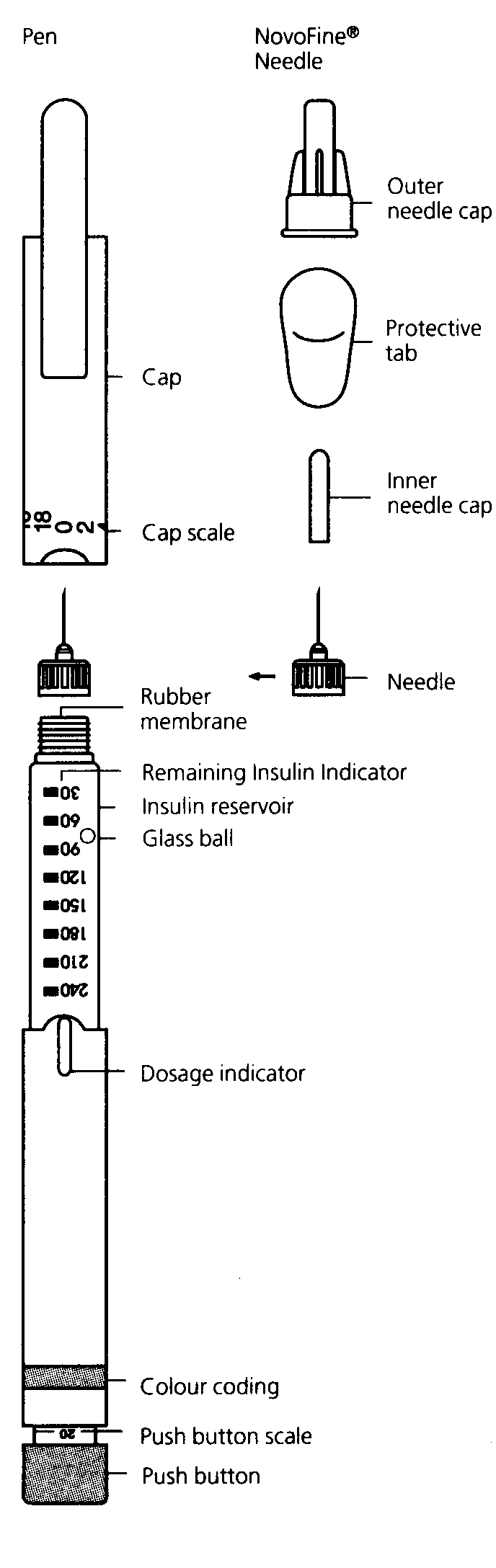
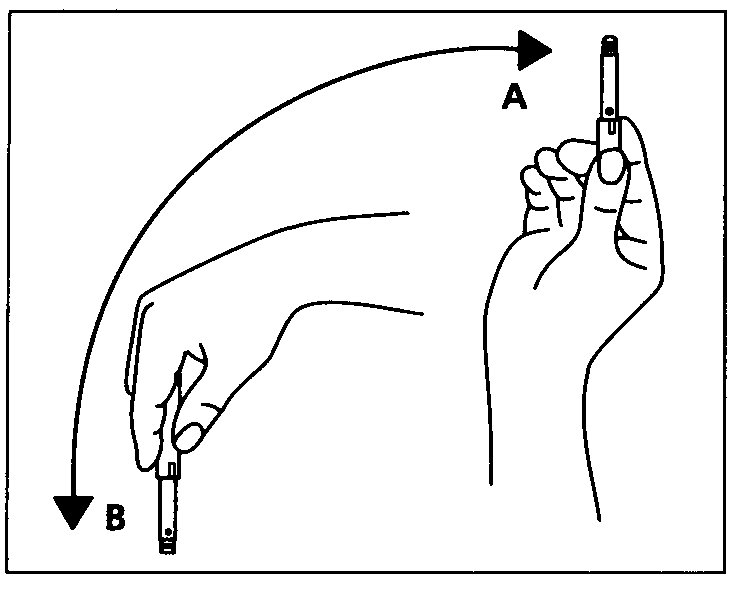
- Preparing the Pen
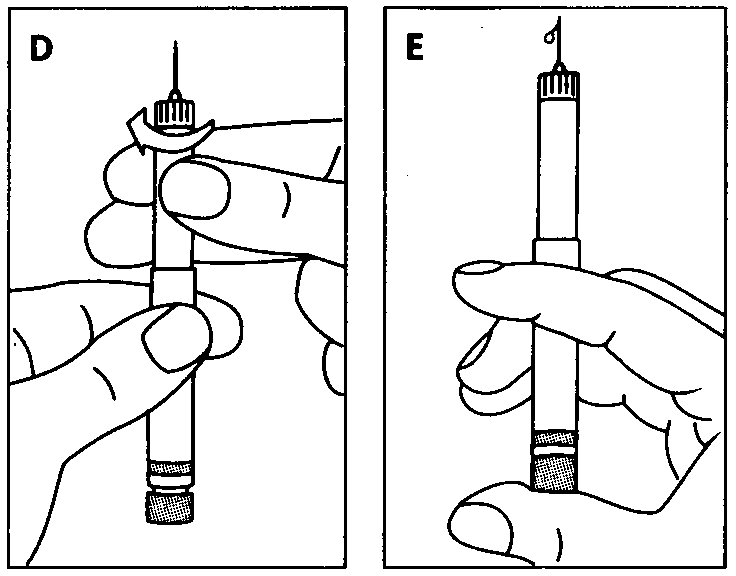
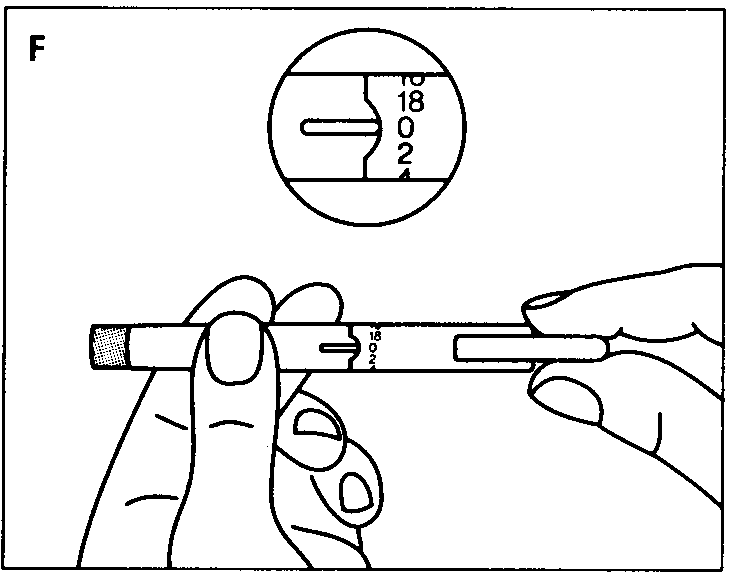
Check that your Pen contains the correct type of insulin. Pull off the cap.
Disinfect the rubber membrane.