Patient Information Leaflet on
‘Penbritin’ paediatric suspension
Please read the leaflet before giving the medicine to your baby
It is essential to follow your doctor's advice. If there is anything you do not understand, please ask your doctor or pharmacist (chemist)
Keep this leaflet until your baby has finished all the prescribed course of 'Penbritin'. You may want to read it again
YOUR BABY'S MEDICINE IS 'PENBRITIN'
What is 'Penbritin'?
It is the maker's name for one of a group of antibiotics called penicillins.
The active ingredient in 'Penbritin' is ampicillin
What does 'Penbritin' do?
It is used to treat a range of bacterial infections
IMPORTANT THINGS TO KNOW ABOUT 'PENBRITIN'
- This medicine is unsuitable for babies who are allergic to penicillin
(You will find more information about this later in the leaflet).
- Give 'Penbritin' to your baby as directed by your doctor. Read the dispensing label.
- It is important to keep giving your baby 'Penbritin' for the length of time your doctor has advised. Do not stop just because your baby seems better.
- Like all medicines, 'Penbritin' can sometimes cause unwanted effects.
(You will find more information about this later in the leaflet).
- Always keep medicines out of the reach of children
NOW TURN OVER FOR DETAILS ON HOW TO GIVE THIS MEDICINE TO YOUR BABY.
BEFORE GIVING YOUR BABY THIS MEDICINE
Is my baby allergic to penicillin?
If you think your baby may be allergic to penicillin, check with your doctor before giving this medicine.
GIVING YOUR BABY THIS MEDICINE
How much should I give my baby and how often?
Follow your doctor's advice about how and when to give your baby
|
‘Penbritin’ paediatric suspension. Doses of ‘Penbritin’ should be taken half to one hour before meals. Please read the dispensing label carefully. Your pharmacist or doctor can help if you are not sure.
How should I give this medicine to my baby?
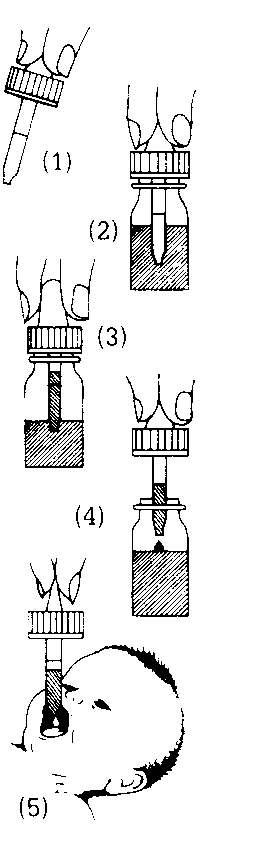
First, shake the bottle. Then:
- (a) If the dropper is not in the bottle:
First remove the cap from the bottle. This cap can be thrown away. Then remove the dropper from its wrapping and squeeze the rubber bulb to expel air (Diagram 1). Follow steps 2, 3, 4, 5 and 6 below.
(b) If the dropper is in the bottle:
Unscrew the dropper from the bottle and squeeze the rubber bulb to expel air (Diagram 1). Follow steps 2, 3, 4, 5 and 6 below.
- Still squeezing the bulb, push the dropper into the medicine in the bottle (Diagram 2).
- Stop squeezing the bulb. The dropper will fill above the line marked on it (Diagram 3).
- Take out the dropper. Squeeze the bulb gently until the medicine is exactly in line with the dose mark (Diagram 4).
- Put the medicine on to the baby's tongue by squeezing the bulb until all the medicine in the dropper has been pushed out (Diagram S). Do not put it on the front of the tongue or it might dribble out of the mouth.
- Wash out the dropper with water. Shake to remove any drops of water. Replace it in the bottle and screw it on tightly.
|
|
What should I do if I forget to give my baby a dose?
If you forget to give your baby a dose at the right time, give it as soon as you remember. Then carry on as before but do not give two doses within an hour or so of each other.
How long should I carry on giving ‘Penbritin’ to my baby?
Follow your doctor’s directions. Do not stop just because your baby seems better. If your baby still seems to be unwell at the end of your prescribed course of treatment, tell your doctor.
AFTER GIVING YOUR BABY THIS MEDICINE
What unwanted effects might it have?
A few people may find that ‘Penbritin’ makes their baby vomit (be sick) or get diarrhoea. If this persists or if the baby is suffering badly, tell your doctor.
Tell your doctor if your baby starts to itch or develops a rash, or any other unusual problems, while you are giving him/her this medicine. If a skin rash occurs, treatment should be stopped.
If your doctor decides to stop the treatment, return any left-over medicine to the pharmacist.
What if I have given my baby too much?
If a large amount has been taken, give plenty of water and contact your doctor or the local hospital casualty department at once.
STORING THIS MEDICINE
How should I store ‘Penbritin’ paediatric suspension?
Keep your baby’s ‘Penbritin’ paediatric suspension in a cool place. Do not freeze. Store all medicines out of the reach of children.
Remember this medicine has been prescribed just for your baby. Do not give this medicine to anyone else – it may not be suitable for them, even if their symptoms seem the same as your baby’s.
MORE INFORMATION ABOUT ‘PENBRITIN’ PAEDIATRIC SUSPENSION
Each 1.25 ml of ‘Penbritin’ paediatric suspension contains 125 mg ampicillin as ampicillin trihydrate.
Non-proprietary name: Ampicillin Paediatric Oral Suspension.
‘Penbritin’ paediatric suspension also contains active ingredients to make the medicine acceptable. Amongst these are sodium benzoate and sucrose.
Manufacturer and Product Licence Holder: Beecham Group plc, Brentford, trading as Beecham Research, Welwyn Garden City, Hertfordshire AL7 1EY.
How can I obtain more information about ‘Penbritin’?
In this short leaflet, we can only give you the most important patient information about ‘Penbritin’. If you would like to know more, ask your doctor or pharmacist.
BEECHAM RESEARCH
‘Penbritin’ is a trade mark.
© 1992 Beecham Research
Leaflet prepared in April 1992
